Subject: FDA
Ricoh's Healthcare Solution Receives FDA 510(k) Clearance for Craniomaxillofacial and Orthopedic Anatomic Modeling
Company takes next major step in providing widespread access to patient-specific 3D models helping healthcare providers deliver precision medicine
EXTON, Pa., June 15, 2022 /PRNewswire/ -- Ricoh USA, Inc. today announced RICOH 3D for Healthcare has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its craniomaxillofacial (CMF) and orthopedic patient-specific anatomic modeling.
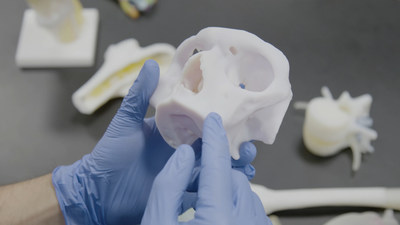
RICOH 3D for Healthcare is an integrated HIPAA-enabled end-to-end workflow solution for the development, design and production of 3D-printed anatomic models, produced using Stratasys 3D printing technology, that enable healthcare providers to develop patient-specific representations of tissue and bone.
The precise replicas help clinicians see inside anatomy for greater visibility into patient needs. RICOH 3D for Healthcare integrates with IBM iConnect® Access, an enterprise imaging solution from IBM Watson Health that is used in many hospitals across the U.S., making it easy to integrate into an existing workflow.
Providers can select from one of two options based on their preferred workstyles or needs. They can choose a point-of-care option, where a Ricoh Managed Services team works onsite at a healthcare facility to manage the entire process using Stratasys 3D printers. Alternatively, healthcare providers can select an on-demand option where they order and have the models printed at a Ricoh facility and shipped directly to them.
"Ricoh is committed to healthcare innovation that will turn the tide on patient engagement and precision medicine. RICOH 3D for Healthcare does just that?offering a model matched to the unique anatomy of each individual patient," said Gary Turner, Managing Director, Additive Manufacturing, North America, Ricoh USA, Inc. "One of our goals is to address the needs of doctors and patients in specific areas with particularly high demand for 3D-printed anatomic models, the 510(k) clearance for CMF and orthopedic models moves us further toward reaching this goal."
According to the Radiological Society of North America (RSNA) 3D Printing Special Interest Group, the use of 3D printing technologies to aid clinical care in the CMF area has been in demand from the advent of 3D printing in the late 1980s. Surgeons, engineers and researchers were designing more manual ways of converting medical imaging datasets into 3D models even before the commercialization of 3D printing technologies.
Patient-specific anatomical models are the baseline in the CMF arena, according to the report, and there is a growing body of work that supports the benefits of 3D-printed models. These include increased comprehension of anatomy and pre-surgical planning for complex surgical cases, as well as a reduction in operation times and improved patient outcomes.
Ricoh plans to solicit 510(k) clearance for additional treatment regions in the future.
For more information about RICOH 3D for Healthcare, view the brochure on the RICOH 3D for Healthcare webpage or follow the company's social media channels on Facebook, Instagram, LinkedIn, Twitter or YouTube.
| About Ricoh |
Ricoh is empowering digital workplaces using innovative technologies and services that enable individuals to work smarter from anywhere.
With cultivated knowledge and organizational capabilities nurtured over its 85-year history, Ricoh is a leading provider of digital services, information management, and print and imaging solutions designed to support digital transformation and optimize business performance.
Headquartered in Tokyo, Ricoh Group has major operations throughout the world and its products and services now reach customers in approximately 200 countries and regions. In the financial year ended March 2022, Ricoh Group had worldwide sales of 1,758 billion yen (approx. 14.5 billion USD).
For further information, please visit www.ricoh.com
© 2022 Ricoh USA, Inc. All rights reserved. All referenced product names are the trademarks of their respective companies.
SOURCE Ricoh USA, Inc.
These press releases may also interest you
|
News published on and distributed by: