Subject: VEN
Subtle Medical Awarded Phase II Funding of $1.6 Million SBIR Grant for Safer MRI Exams and Named to CB Insights Digital Health 150
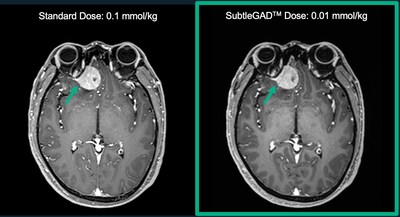
MENLO PARK, Calif., Aug. 13, 2020 /PRNewswire/ -- Subtle Medical is approved for Phase II of its NIH SBIR grant to develop an innovative artificial intelligence (AI)-powered software, SubtleGADtm, which uses deep learning (DL) to reduce gadolinium dosage needed during MRI exams. Phase II funding is based on the results achieved during Phase I, as well as the scientific and technical merit of the project.
SubtleGADtm will benefit millions of patients with advanced chronic kidney disease (CKD) who cannot currently undergo contrast-enhanced MRI, and will reduce the risk of gadolinium retention for many more patients with normal kidney function.
Recognizing the impact of Subtle Medical's AI software on improving patient safety and experience in medical imaging, Subtle Medical was named to 2020 CB Insights Digital Health 150. Subtle was previously named CB Insights Top AI 100.
"Receiving Phase II of the SBIR grant is a tremendous nod to our progress with SubtleGADtm. With this technology, hospitals and imaging centers will be able to deliver safer low-dose contrast-enhanced MRI exams without sacrificing the clinical quality of the images," says Enhao Gong, PhD, Co-founder & CEO. "We appreciate the NIH's support in the development of this novel AI product and look forward to deploying it in clinical settings."
Gadolinium-based contrast agents (GBCAs) are used in approximately one-third of the 40 million MRI scans performed annually. The usage of GBCAs has been linked to the development of nephrogenic systemic fibrosis (NSF), and the recent identification of gadolinium deposition within the brain and body has raised safety concerns about the usage of GBCAs. The FDA has issued warnings about gadolinium retention and required GBCA manufacturers to further assess the safety issues.
Subtle Medical presented SubtleGADtmresearch at RSNA 2018, NeurIPS 2019, and ISMRM 2020, demonstrating positive results from clinical reader studies conducted in multiple institutes in the US and abroad.
SubtleGADtm is the third product under development in Subtle Medical's growing portfolio of AI technologies aimed at improving the quality and efficiency of radiology. SubtleMRtmapplies two methods of image enhancement?denoising and increasing image sharpness?to improve image quality, while SubtlePETtmdenoises scans that were conducted in 25% of the original scan duration. Both products are FDA-cleared and CE-marked to run on all scanner models and are in clinical use in the US and abroad.
About Subtle Medical
Subtle Medical, Inc. is a healthcare technology company with a suite of deep learning solutions that improve workflow efficiency and patient experience. The company has the first AI software solutions FDA-cleared for medical imaging enhancement, SubtlePETtm and SubtleMRtm. Current clinical partners include UCSF, Hoag Hospital, UCSD, Toronto Sickkids, Radnet, DASA, Tiantan Hospital, and Middlesex Hospital, among others. For more information, please visit subtlemedical.com.
Media Contact: Lusi Chien
SOURCE Subtle Medical, Inc.
These press releases may also interest you
|
News published on and distributed by: