Subjects: RCL, CFG
Advisory - Several store-brand pain or sinus relief tablets recalled because consumers may be unable to access important safety information
OTTAWA, Nov. 7, 2018 /CNW/ -
Issue
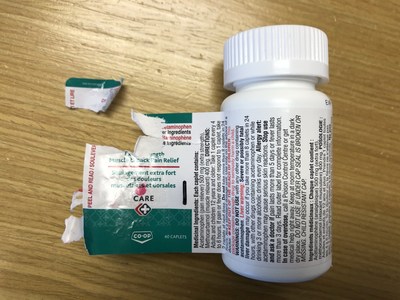
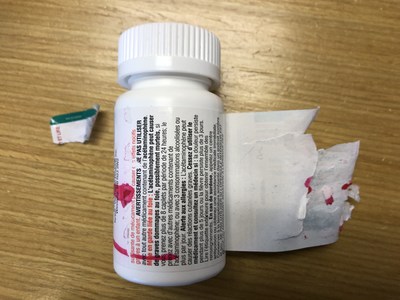
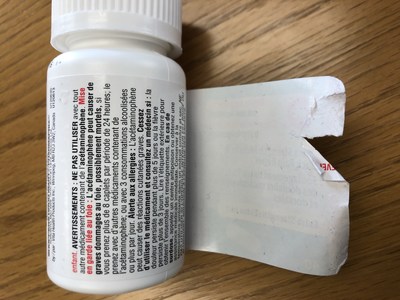
Vita Health Products is voluntarily recalling several store-brand (Care+, Exact, Life, and Pharmasave) over-the-counter drugs used for pain or sinus relief because of a labelling issue. Consumers may be unable to peel open the wrap-around label on the bottle to access the warning statements, or the label may not peel off completely, which may make it difficult to read some of the important safety information.
This issue is the result of a label manufacturing defect affecting some bottles and does not affect the safety or effectiveness of the product in the bottles. The labelling defect means that some consumers may not be able to access important safety information, such as warnings and instructions for product storage.
Vita Health Products has advised Health Canada that only the products listed below are affected by this issue. The products were distributed at major drug and grocery retail chains across Canada.
Who is affected
- Consumers who have bought the affected products listed in the table below.
Affected products
Description | DIN | Lot | Expiry | Units Distributed | |
Care+ brand Extra Strength Muscle & Back Pain Relief | 40 count package | 02239141 | 7L3044GZZ | JL/2020 | 2,973 |
02239141 | 8E2054L0W | NO/2020 | 1,563 | ||
02239141 | 8I1954ML4 | MR/2021 | 1,489 | ||
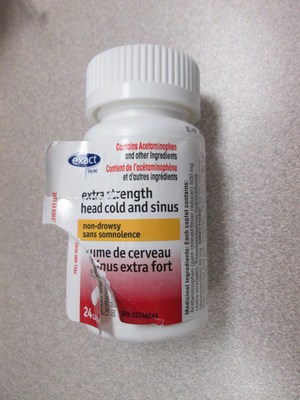
Exact brand Extra Strength Head Cold and Sinus | 24 count package | 02248646 | 8B2924GZW | JA/2021 | 576 |
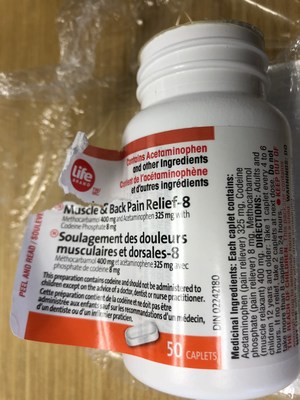
Life brand Muscle & Back Pain Relief-8 | 50 count package | 02242180 | 7J1944M54 | SE/2020 | 3,864 |
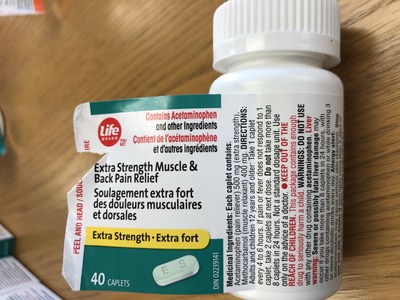
Life brand Extra Strength Muscle & Back Pain Relief | 40 count package | 02239141 | 8E2054HLA | NO/2020 | 7,146 |
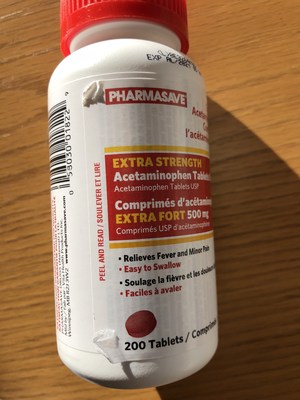
Pharmasave brand Extra Strength Acetaminophen Tablets 500 mg | 200 count package | 00605778 | 8E3264KQS | AL/2021 | 1,022 |
What consumers should do
- Check if your product has a label that does not peel back. If it does not peel back, return the product to the place of purchase, or call the Vita Health Customer Contact Centre at 1?877?637?7557, Monday to Friday 9:00 am to 5:30 pm ET for further instruction.
- Consult your health care professional if you have used these products and have health concerns.
- Report any health product adverse events or complaints to Health Canada.
What Health Canada is doing
Health Canada is monitoring the company's recall. Should additional safety concerns be identified related to this issue, Health Canada will take appropriate action and inform Canadians as necessary.
Stay connected with Health Canada and receive the latest advisories and product recalls.
Également disponible en français

SOURCE Health Canada
These press releases may also interest you
|
News published on and distributed by:



