Subject: WOM
Important Information on MARVELON® 28 (desogestrel/ethinyl estradiol) Oral Contraceptive Tablets - Absence of Day-of-the-week Label Stickers
KIRKLAND, QC, Sept. 11, 2018 /CNW Telbec/ - Merck Canada Inc., known as MSD outside Canada and the United States, notified health care professionals of important information on MARVELON® 28 (desogestrel/ethinyl estradiol) Oral Contraceptive Tablets. The letter stated that some lots of MARVELON® 28 distributed in Canada do not contain day-of-the-week label stickers (see picture below).
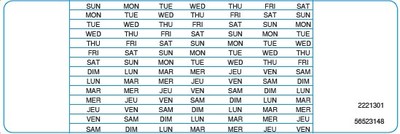
The day-of-the-week label is a sticker that can be applied on the blister pack, indicating the first day of the week when the medication is started. It may help the patient track the tablet intake. The absence of these stickers may increase the risk of non-adherence to the schedule and taking an incorrect number of tablets. Non-adherence to daily oral contraceptive tablet intake can increase the risk of pregnancy. Please note that this issue does not impact the efficacy or safety of the tablet itself if taken as prescribed.
We apologize for this situation and we are working on correcting this issue in a timely manner. In the meantime, we are not distributing MARVELON® 28 until we can start once again delivering product that contain the day-of-the-week label stickers.
Merck Canada Inc. recommends to health care professionals to proactively mention the absence of day-of-the-week label stickers to their patients when prescribing or dispensing MARVELON® 28. Furthermore, patients can be counseled to use a calendar to track the tablet intake.
As of September 10, Merck Canada Inc. will be delivering day-of-the-week label stickers to Canadian retail pharmacies who carry the product in their dispensary as well as clinics which received MARVELON® 28 samples.
We understand that generic alternatives to MARVELON® 28 are also available and do contain day-of-the-week label stickers. We would also like to underline that MARVELON® 21 is not affected by this situation.
Health care professionals or patients do not need to return the MARVELON® 28 products currently on the market, since this issue does not impact the efficacy or safety of the tablet itself if taken as prescribed.
For any questions, patients are instructed to talk to their health care professional.
About MARVELON® (desogestrel/ethinyl estradiol)
To consult the Canadian product monograph for complete prescribing information including contraindications, warnings, precautions, adverse reactions, interactions, dosing and condition of clinical use, please click here.1
About Merck
For over a century, Merck, a leading global biopharmaceutical company known as MSD outside of the United States and Canada, has been inventing for life, bringing forward medicines and vaccines for many of the world's most challenging diseases. Through our prescription medicines, vaccines, biologic therapies and animal health products, we work with customers and operate in more than 140 countries to deliver innovative health solutions. We also demonstrate our commitment to increasing access to health care through far-reaching policies, programs and partnerships. Today, Merck continues to be at the forefront of research to advance the prevention and treatment of diseases that threaten people and communities around the world - including cancer, cardio-metabolic diseases, emerging animal diseases, Alzheimer's disease and infectious diseases including HIV and Ebola. For more information about our operations in Canada, visit www.merck.ca and connect with us on YouTube and Twitter @MerckCanada.
® Merck Sharp & Dohme B.V. Used under license.
Reference: |
|
SOURCE Merck Canada Inc.
These press releases may also interest you
|
News published on and distributed by:



