Subject: RCL
Camber Pharmaceuticals, Inc. Issues Voluntary Nationwide Recall of Atovaquone Oral Suspension, USP 750mg/5mL due to Potential Bacillus cereus Contamination in the Product
Company Contact: Inmar Return Services
Name: Inmar Customer Service - Recalls
Phone Number: 1-877-597-0878
Email: [email protected]
PISCATAWAY, N.J., March 14, 2023 /PRNewswire/ -- Camber Pharmaceuticals, Inc. is voluntarily recalling lot # E220182 of Atovaquone Oral Suspension, USP 750mg/5mL to the Consumer/User level, due to the potential Bacillus cereus contamination in the product.
Risk Statement: In the population most at risk, immunocompromised population, there is a reasonable probability that microbial contamination of Atovaquone Oral Suspension can result in disseminated, life threatening infections such as endocarditis and necrotizing soft tissue infections. To date, Camber has not received any reports of adverse events related to this recall.
Atovaquone Oral Suspension, USP is indicated for prevention and treatment of Pneumocystis jiroveci pneumonia (PCP) in adults and children 13 years of age and older who cannot tolerate other medicines, such as trimethoprim-sulfamethoxazole.
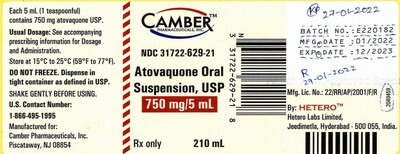
Atovaquone Oral Suspension, USP was distributed Nationwide to Wholesalers, Distributors, Retail Pharmacies, and Mail Order Pharmacies.
The product is packaged in 210mL HDPE bottle in a mono carton. The identified NDC # associated with the product is 31722-629-21, UPC # 331722629218, and the affected lot# is E220182 with an expiration date of 12/2023.
Camber Pharmaceuticals, Inc. is notifying its distributors and customers by our Reverse Logistics Company, Inmar, by mailings and emails communications method and is arranging for returns of all recalled Atovaquone Oral Suspension, USP.
Consumers/distributors/retailers that have product which is being recalled should stop using/return to place of purchase/discard/contact their doctor, etc.
Consumers with questions regarding this recall can contact Inmar by phone at 1-877-597-0878 or email [email protected], Monday ? Friday, 9am ? 5pm Eastern Time. Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to taking or using this drug product.
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- Complete and submit the report Online: www.fda.gov/medwatch/report.htm1
- Regular Mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm2 or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
SOURCE Camber Pharmaceuticals
These press releases may also interest you
|
News published on and distributed by: